
BIO-HELIX - QP019M-0050
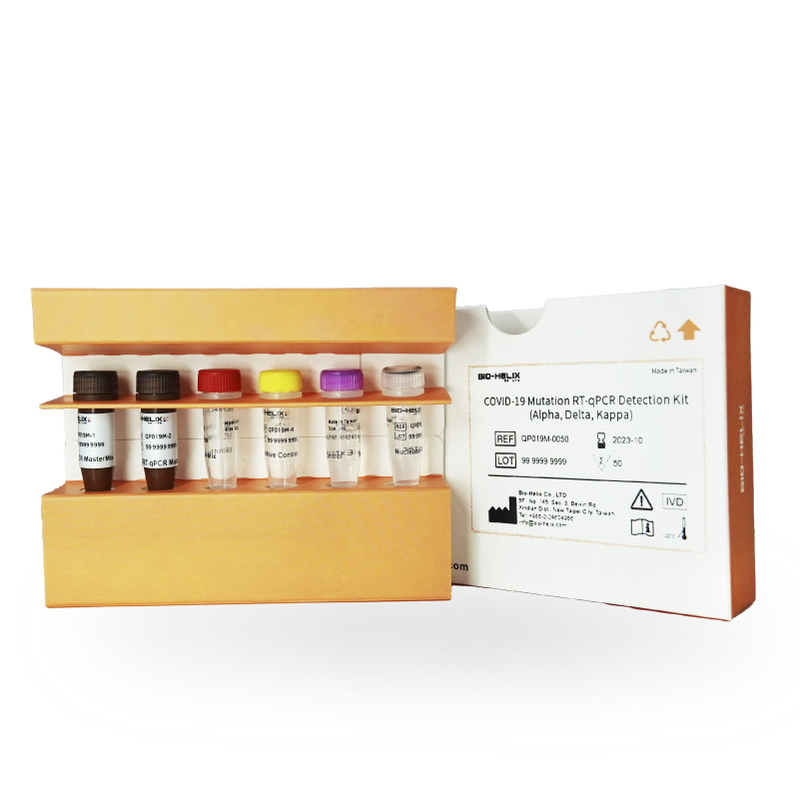
COVID-19 Mutation RT-qPCR Detection Kit (Alpha, Delta, Kappa)
Size: 50 Rxns
Since the onset of the COVID-19 pandemic in early 2020, new genetic variants of concern have been identified and reported in different geographical regions. Genetic changes in the SARS-CoV-2 virus, as universally understood, are significantly associated with its mutability during viral replication and rendering the altered SARS-CoV-2 genetic variants with a competitive advantage. The conferred advantages include but are not limited to conformational changes in the virus spike protein allowing evasion of immune responses and enhanced transmissibility due to stronger binding to target viral ACE2 receptors, for example, on the surface of respiratory tract cells, allowing enhanced entry into the target cell. As a result, the Alpha, Delta, and Kappa variants have spread rapidly across the globe creating concern as to their transmissibility and impact on current vaccines, severity of hospitalization, and degree of mortality.
For advancing the knowledge and competency in assessing the impact and dynamics of the SARS-CoV-2 pandemic, we are committed to providing the research communities across the globe with the highest possible quality, Research-Use-Only molecular assay kit designed for the in vitro qualitative detection and differentiation of the mutations in N501Y (Alpha Type) and L452R (Delta/Kappa Type) present in the nasal and nasopharyngeal swab specimens from patients confirmed with known SARS-CoV-2 infection.

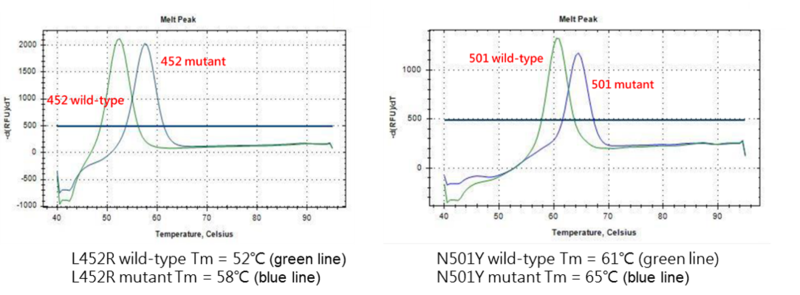

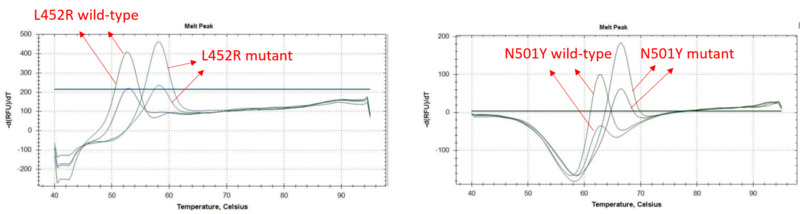
A study was performed to assess the limit of detection performance of COVID-19 Mutation RT-qPCR Detection Kit (Alpha, Delta, Kappa). A testing concentration were carried out with 1000 and 200 copies SARS-CoV-2 RNA per reaction separately and tested on QuantStudio™ 5 Real-Time PCR System. The synthetic SARS-CoV-2 RNA were obtained from Twist Biosciences (Assay Ready Control 14,EPI_ISL_710528 and Synthetic RNA Control 18, EPI_ISL_1662307).
| Name | Download |
|---|---|
| PROTOCOL | QP019M_Protocol_v1_20211019.pdf |
| Safety Data Sheet|SDS | QP019M-0500_COVID-19_Mutation_RT-qPCR_Detection_Kit__Alpha__Delta__Kappa__SDS.pdf |